Transcatheter Edge-to-Edge Repair (TEER):Once Again Upgraded for SMR† patients
2022 AHA/ACC/HFSA Guideline1 for the Management of Patients with Heart Failure strengthen their recommendation of TEER for COAPT-like* secondary MR patients to a Class 2a.
† Secondary MR patients despite optimal GDMT.
*COAPT-like: NYHA II-IV; severe secondary MR; suitable anatomy; LVEF 20%-50%; LVESD ≤70 mm; PASP ≤70 mm Hg.
- Heidenreich PA, et al. "2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association. Joint Committee on Clinical Practice Guidelines." J Am Coll Cardiol. Apr 01,2022. Epublished DOI: 10.1016/j.jacc.2021.12.012.
† Secondary MR patients despite optimal GDMT.
*COAPT-like: NYHA II-IV; severe secondary MR; suitable anatomy; LVEF 20%-50%; LVESD ≤70 mm; PASP ≤70 mm Hg.
- Heidenreich PA, et al. "2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association. Joint Committee on Clinical Practice Guidelines." J Am Coll Cardiol. Apr 01,2022. Epublished DOI: 10.1016/j.jacc.2021.12.012.


MitraClip is the First and Only TMVr* therapy shown to improve survival in secondary MR2,3
- Dramatic improvement in health-related quality of life4
- MitraClip patients were 2.5 times more likely to experience a large improvement in Kansas City Cardiomyopathy Questionnaire (KCCQ) score compared to patients receiving standard care4
SEE MITRACLIP’S DATA ON TREATING SECONDARY MITRAL REGURGITATION
- Reduction in heart failure (HF) hospitalization risk3
- In the COAPT™ trial, MitraClip reduced the relative risk of HF hospitalization by 47% at 2 years and 51% at 3 years3
*TMVr is now also referred to as TEER (transcatheter edge-to-edge repair)
TRANSCATHETER MITRAL VALVE REPAIR (TMVr) IS A MINIMALLY INVASIVE PROCEDURE
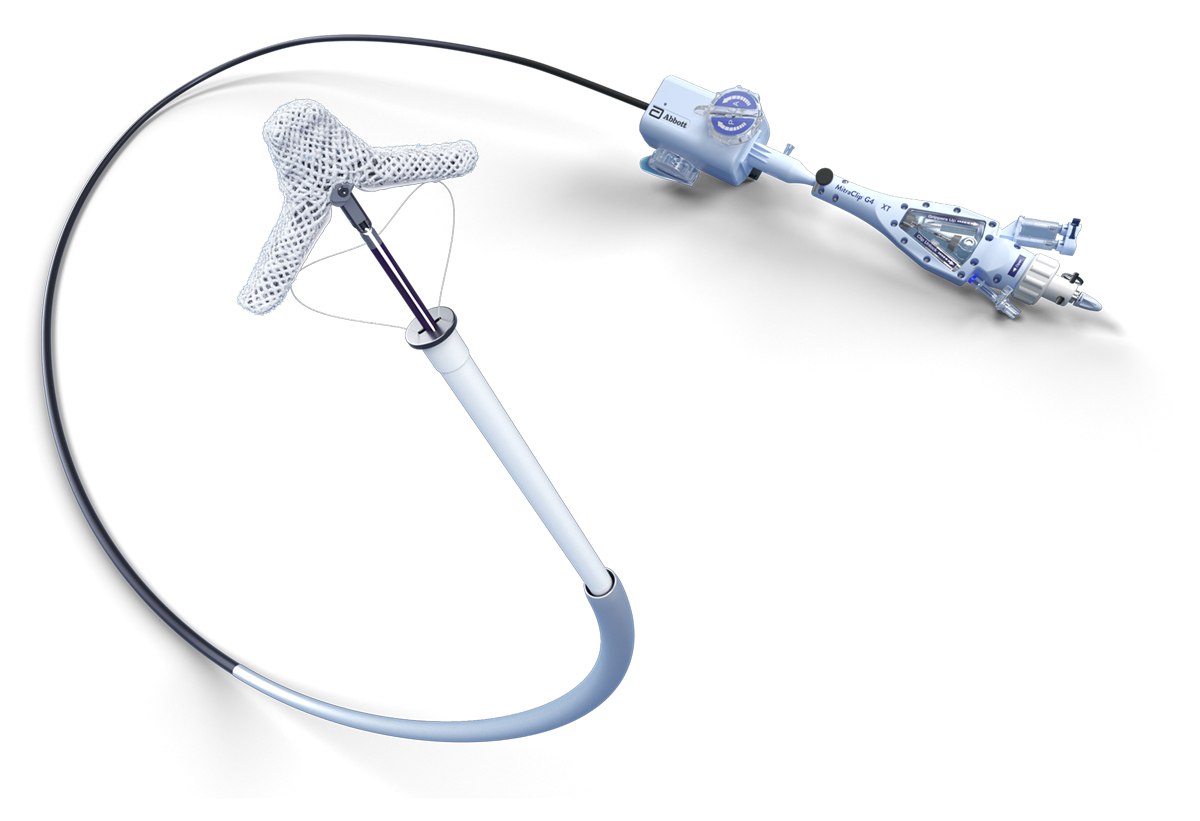
The MitraClip procedure is a minimally invasive, catheter-based therapy with over 17+ years of clinical use. It is performed by multidisciplinary heart teams using venous access and real-time imaging (echocardiography and fluoroscopy), thereby avoiding cardiopulmonary bypass. The MitraClip device grasps and coapts the mitral valve leaflets, resulting in fixed coaptation (approximation) of the mitral leaflets throughout the cardiac cycle.5
Durable outcomes in prohibitive risk patients with primary (degenerative) MR6
- The power to improve cardiac function and achieve reverse remodeling
- Substantial improvement New York Heart Association (NYHA) class maintained over the long term
SEE THE DATA ON TREATING PRIMARY MITRAL REGURGITATION
MAT-2410455 v1.0 | Item approved for U.S. use only
- Rottbauer WD. Contemporary Clinical Outcomes with MitraClip™ (NTR/XTR) System: Core-lab Echo Results from +1000 Patient the Global EXPAND Study. Data presented at PCR 2020.
- Stone GW, Lindenfeld J, Abraham WT, et al. Transcatheter mitral-valve repair in patients with heart failure. N Engl J Med. 2018;379(24):2307-2318.
- Mack M. COAPT: 3-Year Outcomes of Transcatheter Mitral Valve Repair in Patients with Heart Failure. JACC Mar 2021.
- Arnold SV, Chinnakondepalli KM, Spertus JA, et al. Health status after transcatheter mitral valve repair in heart failure and secondary mitral regurgitation. COAPT Trial. J Am Coll Cardiol. 2019;73(17):2123-2132.
- MitraClip™ Clip Delivery System Instructions for Use.
- Kar S, Feldman T, Qasim A, et al. Five-year outcomes of transcatheter reduction of significant mitral regurgitation in high-surgical risk patients. Heart. 2019;105:1622-1628.
REQUEST AN ABBOTT REP
Rx Only
Important Safety Information
MITRACLIP™ CLIP DELIVERY SYSTEM
- The MitraClip™ G4 System is indicated for the percutaneous reduction of significant symptomatic mitral regurgitation (MR ≥ 3+) due to primary abnormality of the mitral apparatus [degenerative MR] in patients who have been determined to be at prohibitive risk for mitral valve surgery by a heart team, which includes a cardiac surgeon experienced in mitral valve surgery and a cardiologist experienced in mitral valve disease, and in whom existing comorbidities would not preclude the expected benefit from reduction of the mitral regurgitation.
- The MitraClip™ G4 System, when used with maximally tolerated guideline-directed medical therapy (GDMT), is indicated for the treatment of symptomatic, moderate-to-severe or severe secondary (or functional) mitral regurgitation (MR; MR ≥ Grade III per American Society of Echocardiography criteria) in patients with a left ventricular ejection fraction (LVEF) ≥ 20% and ≤ 50%, and a left ventricular end systolic dimension (LVESD) ≤ 70 mm whose symptoms and MR severity persist despite maximally tolerated GDMT as determined by a multidisciplinary heart team experienced in the evaluation and treatment of heart failure and mitral valve disease.
Contraindications
The MitraClip G4 System is contraindicated in patients with the following conditions: Patients who cannot tolerate, including allergy or hypersensitivity to, procedural anticoagulation or post procedural anti-platelet regime; Patients with known hypersensitivity to clip components (nickel / titanium, cobalt, chromium, polyester), or with contrast sensitivity; Active endocarditis of the mitral valve; Rheumatic mitral valve disease; Evidence of intracardiac, inferior vena cava (IVC) or femoral venous thrombus.
Potential Complications and Adverse Events
The following ANTICIPATED EVENTS have been identified as possible complications of the MitraClip G4 procedure: Allergic reactions or hypersensitivity to latex, contrast agent, anaesthesia, device materials (nickel / titanium, cobalt, chromium, polyester), and drug reactions to anticoagulation, or antiplatelet drugs, Vascular access complications which may require transfusion or vessel repair including: wound dehiscence, catheter site reactions, Bleeding (including ecchymosis, oozing, hematoma, hemorrhage, retroperitoneal hemorrhage), Arteriovenous fistula, pseudoaneurysm, aneurysm, dissection, perforation / rupture, vascular occlusion, Emboli (air thrombotic material, implant, device component); Peripheral Nerve Injury; Lymphatic complications; Pericardial complications which may require additional intervention, including: Pericardial effuse on, Cardiac tamponade, Pericarditis; Cardiac complications which may require additional interventions or emergency cardiac surgery, including: Cardiac perforation, Atrial septal defect; Mitral valve complications, which may complicate or prevent later surgical repair, including: Chordal entanglement / rupture, Single Leaflet Device Attachment (SLDA), Thrombosis, Dislodgement of previously implanted devices, Tissue damage, Mitral valve stenosis, Persistent or residual mitral regurgitation, Endocarditis; Cardiac arrhythmias (including conduction disorders, atrial arrhythmias, ventricular arrhythmias); Cardiac ischemic conditions (including myocardial infarction, myocardial ischemia, and unstable / stable angina); Venous thromboembolism (including deep vein thrombosis, pulmonary embolism, post procedure pulmonary embolism); Stroke / Cerebrovascular accident (CVA) and Transient Ischemic Attack (TIA); System organ failure: Cardio-respiratory arrest, Worsening heart failure, Pulmonary congestion, Respiratory dysfunction / failure / atelectasis, Renal insufficiency or failure, Shock (including cardiogenic and anaphylactic); Blood cell disorders (including coagulopathy, hemolysis, and Heparin Induced Thrombocytopenia (HIT)); Hypotension / hypertension; Infection including: Urinary Tract Infection (UTI), Pneumonia, Septicemia; Nausea / vomiting; Chest pain; Dyspnea; Edema; Fever or hyperthermia; Pain; Death; Fluoroscopy, Transesophageal echocardiogram (TEE) and Transthoracic echocardiogram (TTE) -related complications: Skin injury or tissue changes due to exposure to ionizing radiation, Esophageal irritation; Esophageal perforation, Gastrointestinal bleeding.
